 Further, each carbon atom that supports a hydroxyl group (except for the first and last) is chiral, giving rise to a number of isomeric forms all with the same chemical formula. Monosaccharides are the building blocks of disaccharides like sucrose (common sugar) and polysaccharides (such as cellulose and starch). Examples of monosaccharides include glucose (dextrose), fructose, galactose, xylose and ribose. 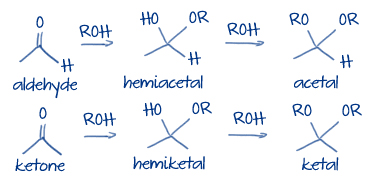
They consist of one sugar and are usually colorless, water- soluble, crystalline solids. They cannot be hydrolyzed into simpler sugars. Monosaccharides (from Greek monos: single, sacchar: sugar) are the simplest carbohydrates. Risk calculators and risk factors for Monosaccharide US National Guidelines Clearinghouse on Monosaccharideĭirections to Hospitals Treating Monosaccharide Ongoing Trials on Monosaccharide at Clinical Ĭlinical Trials on Monosaccharide at Google Mutarotation: The change between two anomers in solution when the corresponding stereocenters interconvert.Articles on Monosaccharide in N Eng J Med, Lancet, BMJ Β form: The configuration of a cyclic monosaccharide where the oxygen attached to the anomeric carbon is on the same side of the ring as the CH₂ OH group. Α form: The configuration of a cyclic monosaccharide where the oxygen attached to the anomeric carbon is on the opposite side of the ring from the CH₂ OH group. Stereoisomers: Each of two or more compounds differing only in the spatial arrangement of their atoms.Īnomers: A type of stereoisomer that differs in configuration at the hemiacetal or acetal carbon they are a specific type of epimer.Īnomeric carbon: A carbon derived from the carbonyl carbon (the ketone or aldehyde functional group) of the open-chain form of the carbohydrate molecule. Haworth projection: A simple three-dimensional representation of a monosaccharide. Monosaccharides: The basic unit of carbohydrates that cannot be hydrolyzed to simpler chemical compounds with the general formula (CH 2O) n.Īqueous solutions: Solutions in which the solvent is water.įischer projection: A two-dimensional representation of a molecule that maintains information about its absolute configuration. In an aqueous solution, an equilibrium mixture forms between the two anomers and the straight-chain structure of a monosaccharide in a process known as mutarotation.Two cyclic stereoisomers can form from each straight-chain monosaccharide these are known as anomers.Monosaccharides that contain five or more carbons atoms form cyclic structures in aqueous solutions.Cyclic compounds undergo mutarotation, in which they shift from one anomeric form to another with the straight-chain structure. The opening and closing of sugars repeats continuously in an ongoing interconversion between anomeric forms and is referred to as mutarotation. When the hydroxyl is down, glucose is said to be in its α form, and when it’s up, glucose is said to be in its β form. During ring formation, the O from the carbonyl, which is converted to a hydroxyl group, will be trapped either “above” the ring (on the same side as the CH₂ OH) or “below” the ring (on the opposite side from this group). 
The anomeric carbon can take on either an α- or β-conformation when the ring forms. When a straight-chain monosaccharide forms a cyclic structure, the carbonyl oxygen atom may be pushed either up or down, giving rise to two stereoisomers that are anomers. In the figure below, you can see (a) the two-dimensional Fischer projection of glucose, (b) a three-dimensional representation of the glucose molecule during ring formation, and (c) the final cyclic form of glucose in simple three-dimensional representation (called a Haworth projection). These forms exist in equilibrium with each other, but equilibrium strongly favors the ring forms (particularly in aqueous, or water-based, solution). Many five- and six-carbon sugars can exist either as a linear chain or in one or more ring-shaped forms. Monosaccharides that contain five or more carbon atoms form cyclic structures in aqueous solutions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
